CLINICAL TRIAL DATA
ZILBRYSQ delivered statistically significant and sustained improvements in activities of daily living for adults with anti-AChR Ab+ gMG1,2

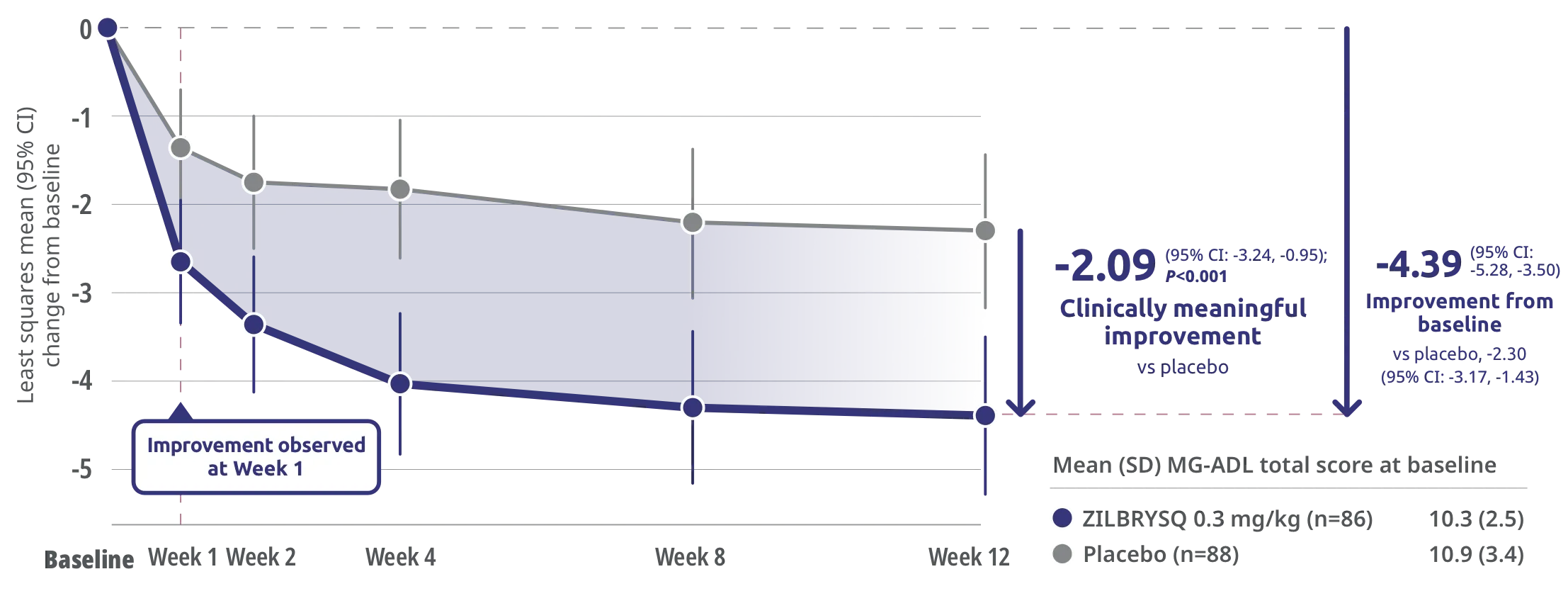
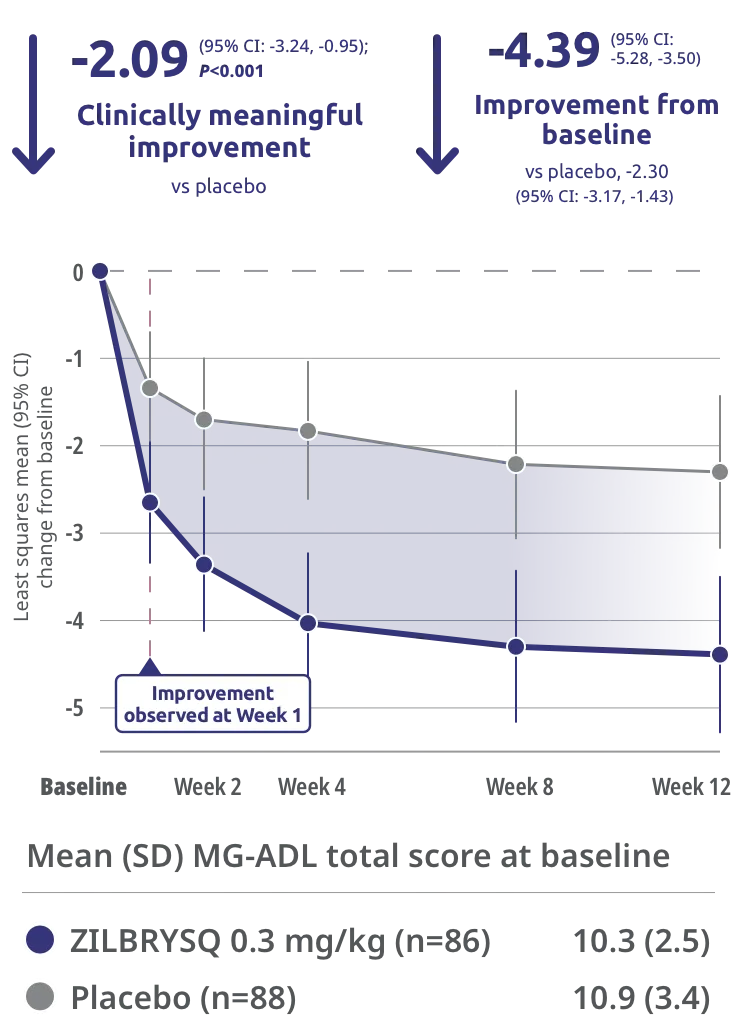
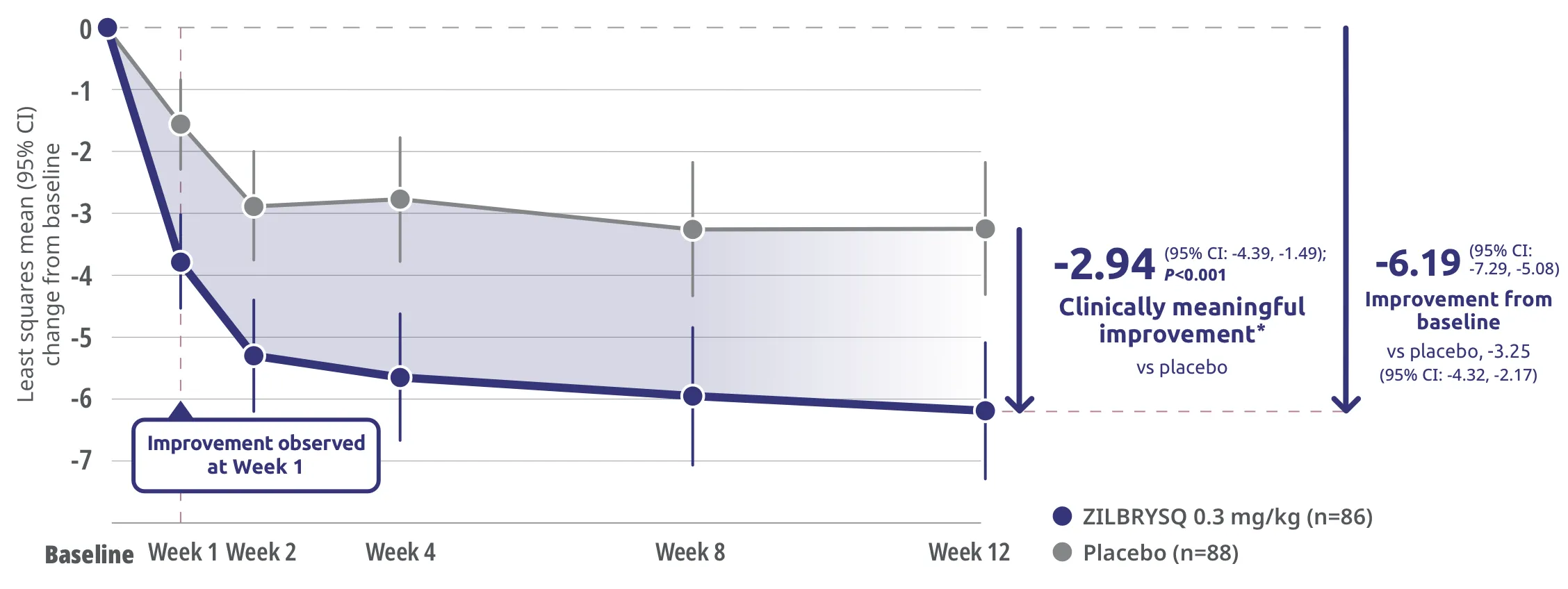
RAISE: Primary Endpoint MG-ADL
Rapid and statistically significant improvements at Week 121,3
In the pivotal Phase 3 RAISE trial, ZILBRYSQ delivered a >4-point improvement in the ability to manage activities of daily living at Week 12 for adults with anti-acetylcholine receptor (AChR) antibody positive (Ab+) generalized myasthenia gravis (gMG).
Primary endpoint: Change from baseline (CFB) at Week 12 in Myasthenia Gravis Activities of Daily Living (MG-ADL) total score
The most common adverse reactions (reported in at least 10% of patients treated with ZILBRYSQ) were injection site reactions, upper respiratory tract infections, and diarrhea.1
Clinically meaningful was defined as a ≥2-point change in MG-ADL score.3
CI=confidence interval.
The safety and efficacy of ZILBRYSQ were evaluated in a 12-week, multicenter, randomized, double-blind, placebo-controlled study. Patient population included adult patients with diagnosis of mild to severe gMG (MGFA class II-IV).1
"This kind of potential for meaningful improvement in only 3 months is very appealing to me when I am considering ZILBRYSQ for my patients.”
Watch Dr. Michael Weiss present the RAISE and RAISE-XT clinical trials
Myasthenia Gravis Activities of Daily Living (MG-ADL)
Assesses the impact of gMG on daily functions of 8 symptoms on a scale of 0-3, with total scores ranging from 0 to 24. Higher scores are interpreted as greater impairments.1
- An improvement of ≥2 points was established as clinically meaningful.3
Measures include4:
- Talking
- Chewing
- Swallowing
- Breathing
- Brushing teeth and/or combing hair
- Rising from a chair
- Diplopia
- Eyelid droop
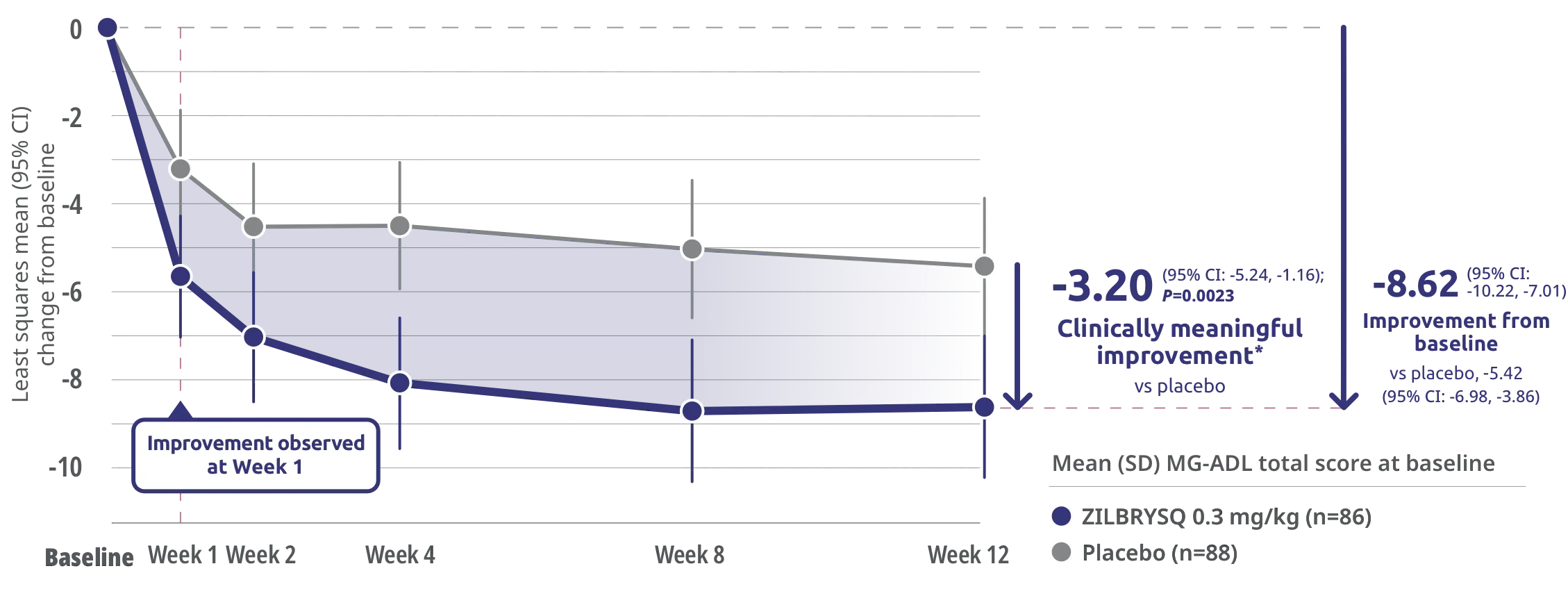
RAISE-XT: Secondary Endpoint MG-ADL
Sustained efficacy through Week E842,5
The primary endpoint of RAISE-XT evaluated the long-term safety and tolerability of ZILBRYSQ. Please see the results here.
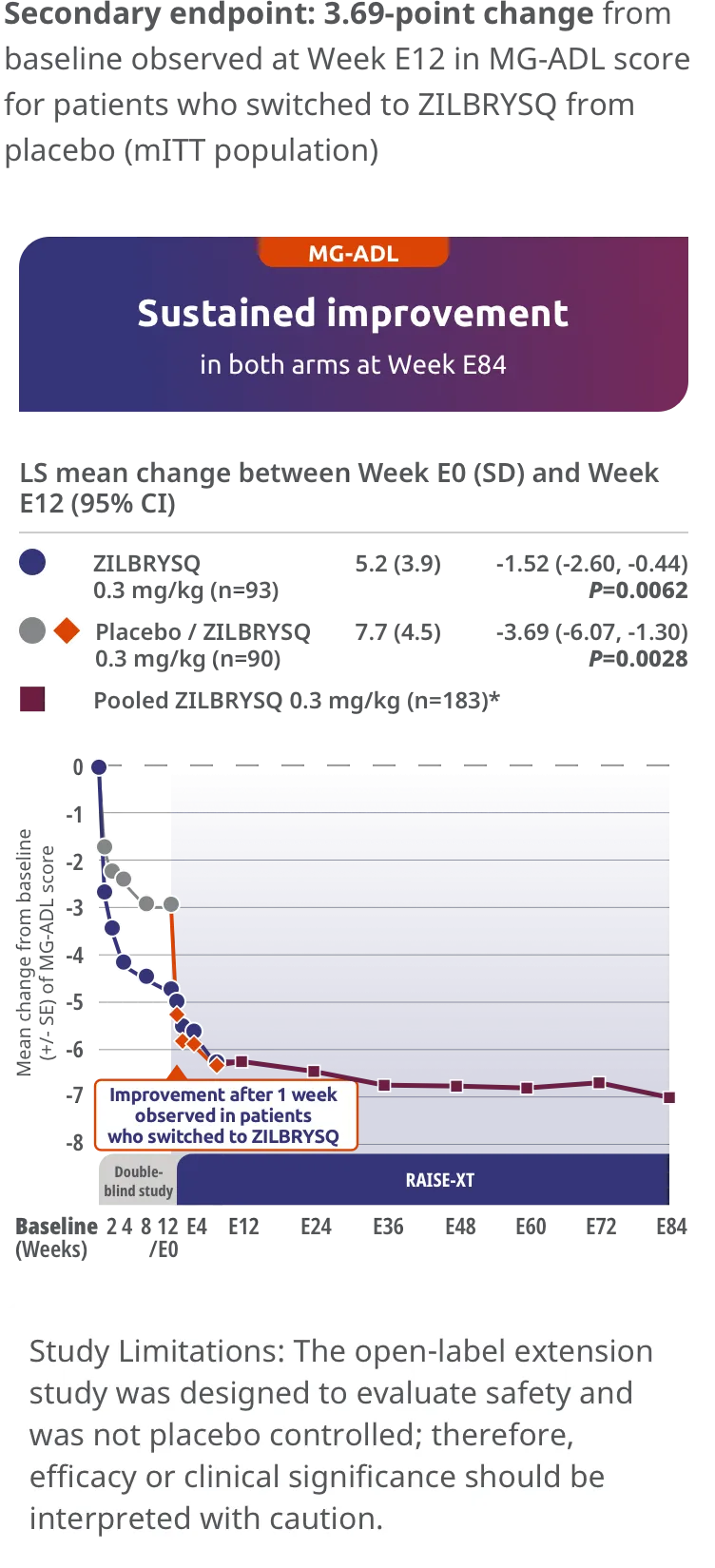
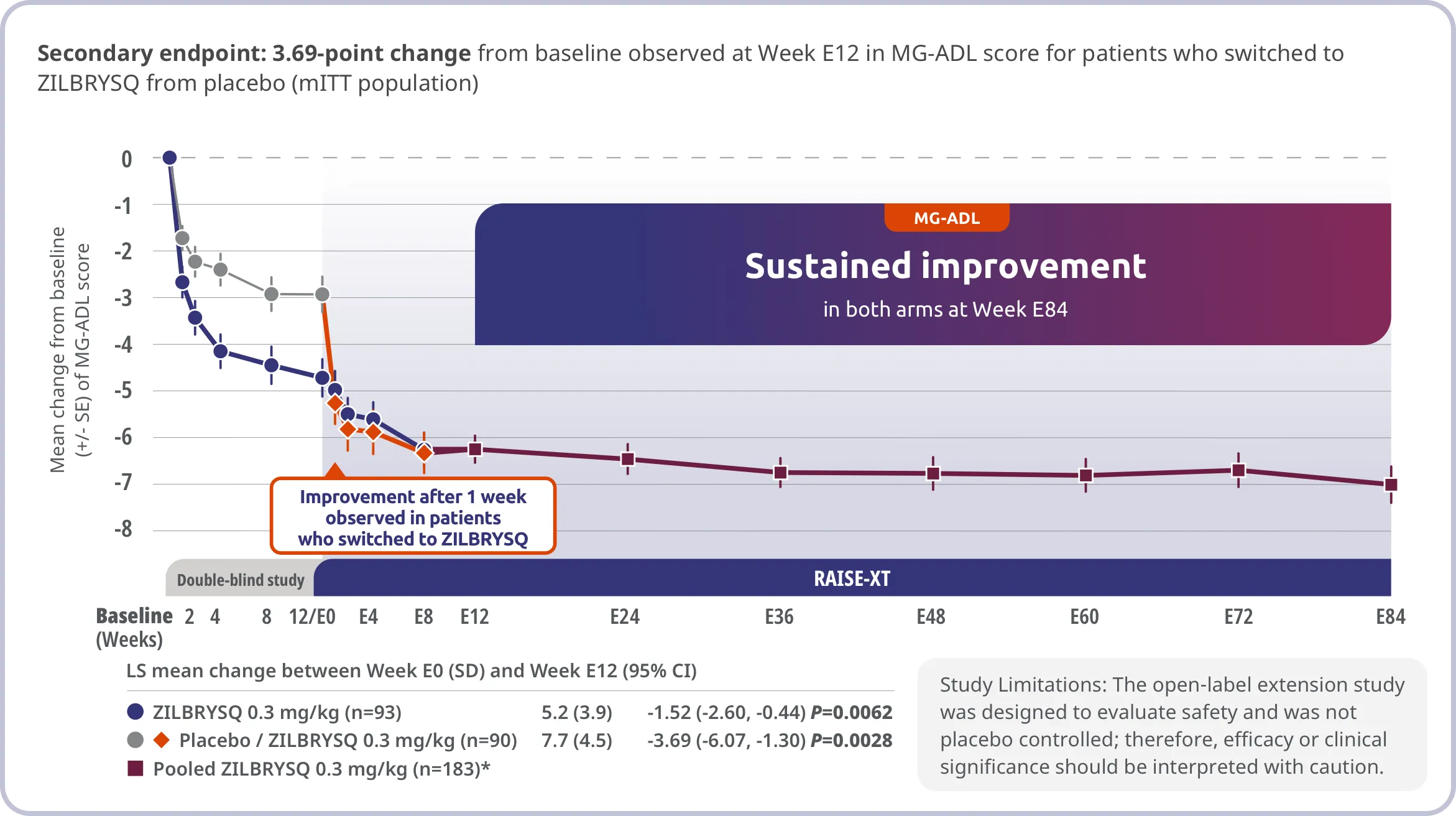
The long-term safety, tolerability, and efficacy were evaluated in an open-label extension study comprised of gMG patients who completed either Phase 2 or Phase 3 (RAISE) of the clinical trials.5
RAISE-XT evaluated 200 adult patients from either Phase 2 or 3, including 17 patients from Phase 2 who started with or switched to 0.1 mg/kg. The graphic above shows only the 183 patients continuing or switching to ZILBRYSQ at the approved dosage (0.3 mg/kg) and are solely represented in the subsequent analyses on efficacy.2,5
*Includes pooled data of patients from Week E12-Week E84.2
CI=confidence interval; LS=least squares; MGFA=Myasthenia Gravis Foundation of America; mITT=modified intention-to-treat; SE=standard error.
MG-ADL responder rates for ZILBRYSQ in RAISE and RAISE-XT1,2
A high proportion of patients taking ZILBRYSQ were MG-ADL clinical responders (≥ a 3-point improvement from baseline) at Week 12 and Week E84.1,2
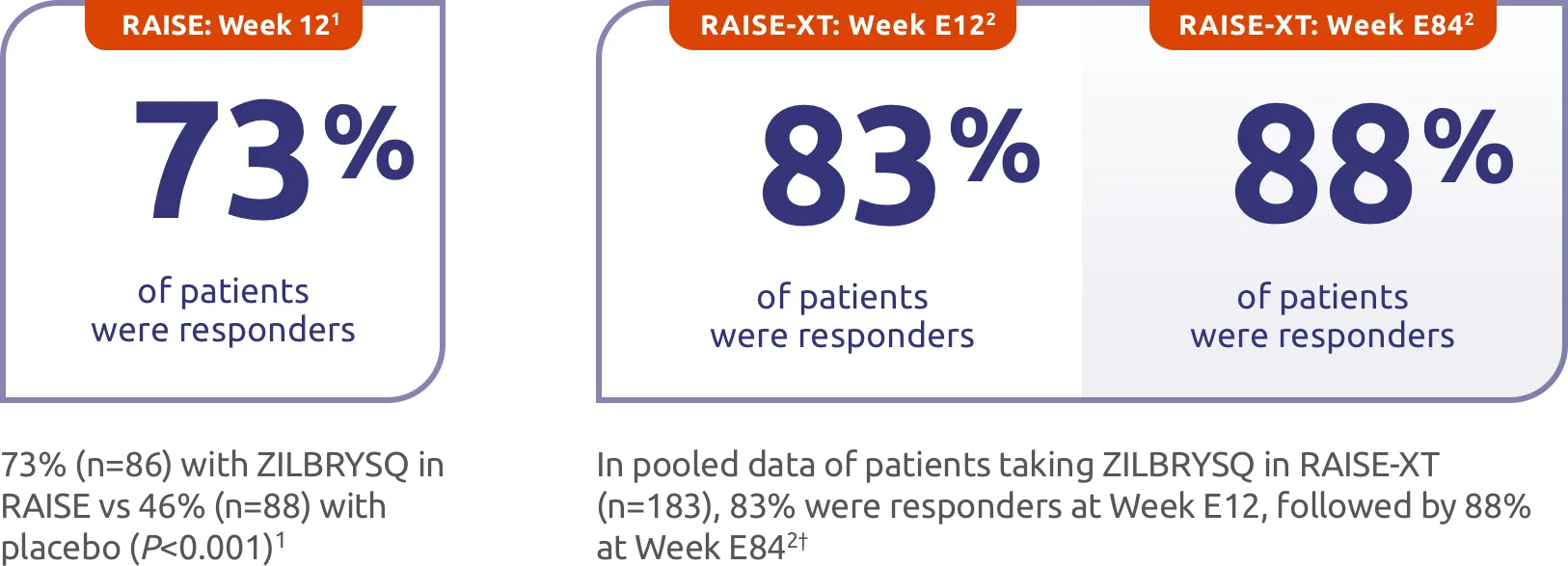
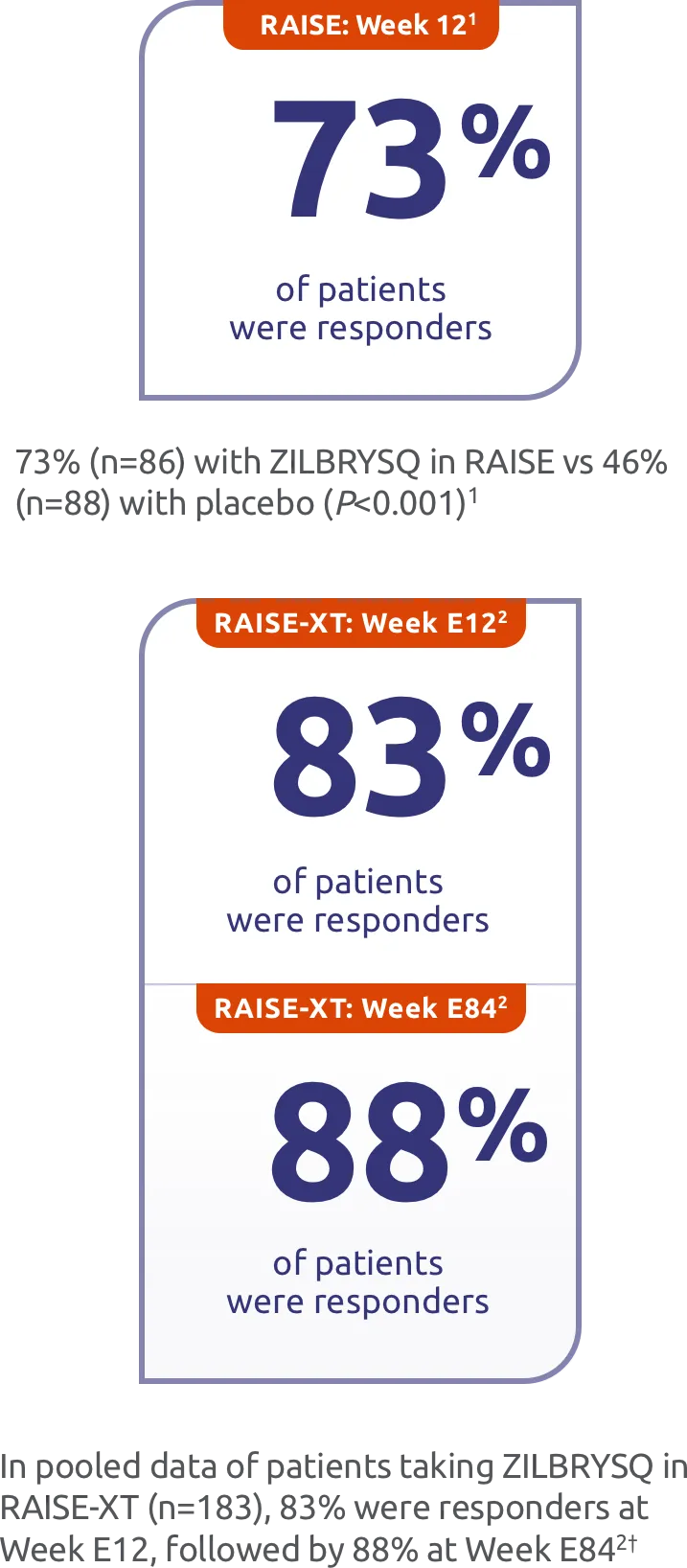
Responder rate was specified as an other secondary efficacy endpoint in the RAISE study and an exploratory efficacy endpoint in RAISE-XT. Results should be interpreted with caution.3,5
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
Includes pooled data of patients from Phases 2 and 3 of the clinical study who either continued on ZILBRYSQ or switched to ZILBRYSQ from placebo.2
ZILBRYSQ showed continued minimal symptom expression (MSE) in patients through Week E842
Patients who achieved MSE in either RAISE or RAISE-XT achieved an MG-ADL score of 0 or 1 without rescue therapy.5
Patients taking ZILBRYSQ who achieved MSE (mITT population)
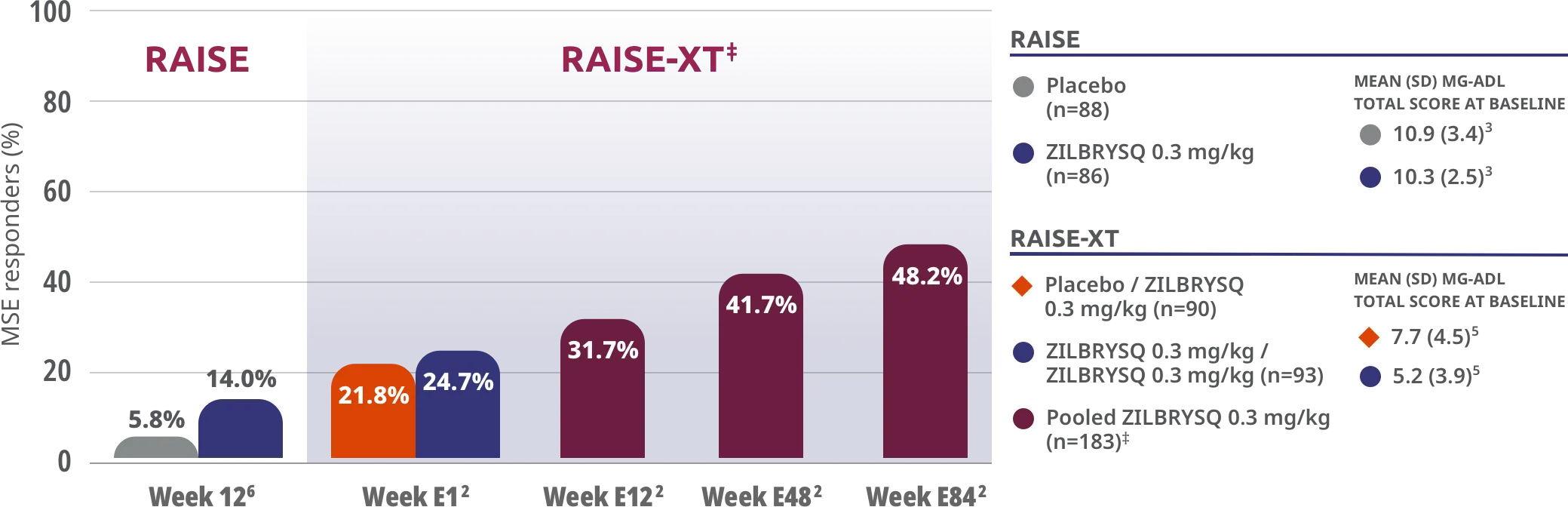
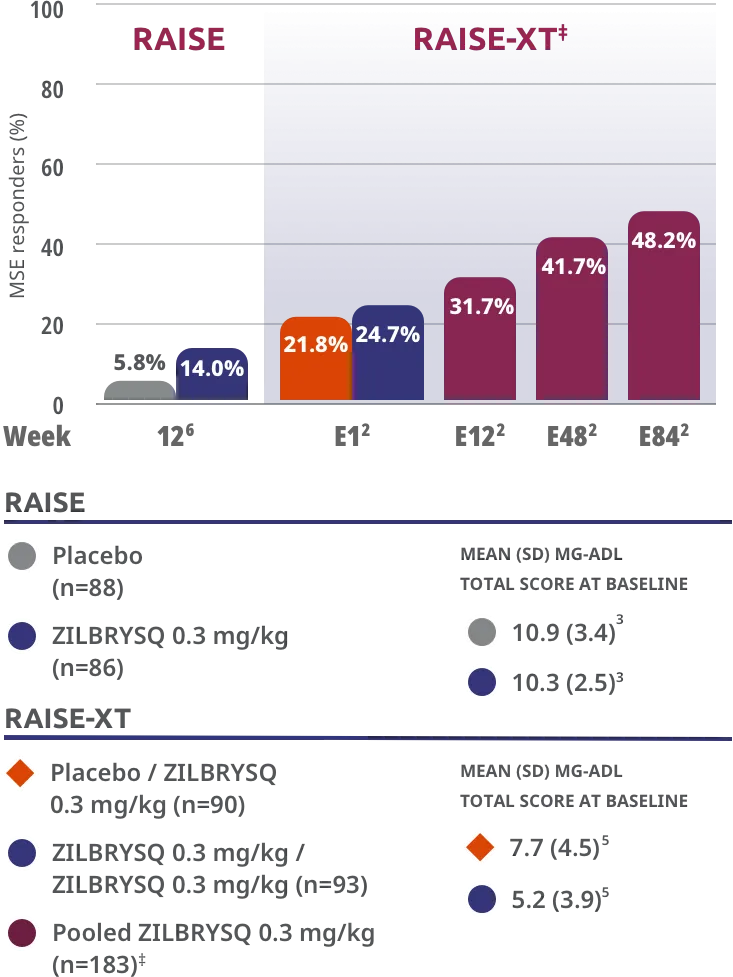
MSE was defined as an MG-ADL total score of 0-1. MSE was specified as an other secondary efficacy endpoint in the RAISE study and an exploratory endpoint in RAISE-XT. Results should be interpreted with caution.3,5
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
Includes pooled data of patients from Week E12-Week E84.2
RAISE-XT evaluated 200 patients from either Phase 2 or 3, including 17 patients from Phase 2 who started with or switched to 0.1 mg/kg. The graphic above shows only the 183 patients continuing or switching to ZILBRYSQ at the approved dosage (0.3 mg/kg) and are solely represented in the subsequent analyses on efficacy.2,5
mITT=modified intention-to-treat; SD=standard deviation.
The efficacy and safety of ZILBRYSQ for the treatment of generalized myasthenia gravis (gMG) in adult patients who are anti-acetylcholine receptor (AChR) antibody positive (Ab+) were established in RAISE, a 12-week, multicenter, randomized, double-blind, placebo-controlled study. A total of 174 patients were randomized to receive either ZILBRYSQ (n=86) or placebo (n=88).1,3
RAISE secondary endpoints
RAISE: consistent and significant improvements across key clinician- and patient-reported secondary and other outcome measures1,3
Statistically significant improvement in muscle strength1,3
Secondary endpoint: CFB in QMG score at Week 12
The safety and efficacy of ZILBRYSQ were evaluated in a 12-week, multicenter, randomized, double-blind, placebo-controlled study. Patient population included adult patients with diagnosis of mild to severe gMG (MGFA class II-IV).1
*Clinically meaningful was defined as a ≥3-point change in QMG score.3
CFB=change from baseline; CI=confidence interval; MGFA=Myasthenia Gravis Foundation of America.
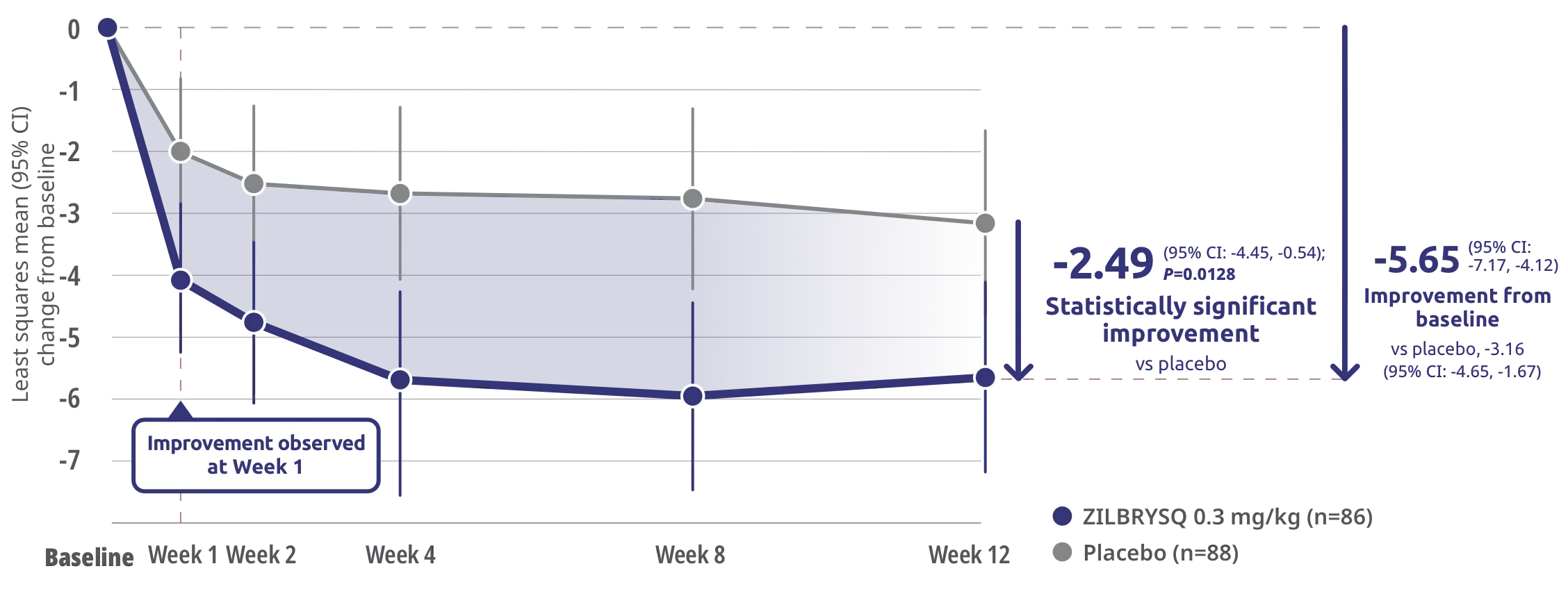
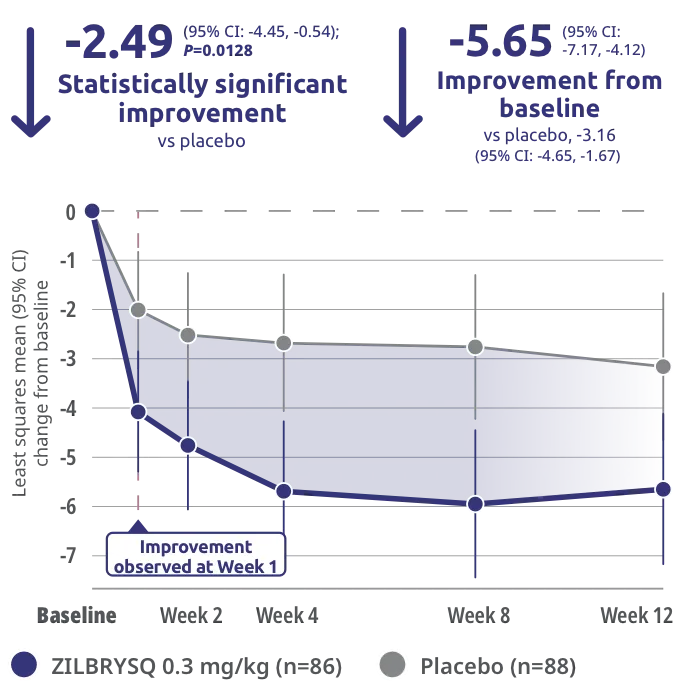
Statistically significant improvement in gMG signs and symptoms3
Secondary endpoint: CFB in MGC score at Week 123
The safety and efficacy of ZILBRYSQ were evaluated in a 12-week, multicenter, randomized, double-blind, placebo-controlled study. Patient population included adult patients with diagnosis of mild to severe gMG (MGFA class II-IV).1
*Clinically meaningful was defined as a 3-point change in MGC score.3
CFB=change from baseline; CI=confidence interval; MGFA=Myasthenia Gravis Foundation of America.
Statistically significant improvements in quality of life3
Secondary endpoint: CFB in MG-QoL 15r score at Week 123
The safety and efficacy of ZILBRYSQ were evaluated in a 12-week, multicenter, randomized, double-blind, placebo-controlled study. Patient population included adult patients with diagnosis of mild to severe gMG (MGFA class II-IV).1
Study Limitation: At the time of the study, no threshold for clinical meaningfulness for the MG-QoL 15r assessment had been established.3
CFB=change from baseline; CI=confidence interval; MGFA=Myasthenia Gravis Foundation of America.
RAISE secondary endpoint measures overview
Quantitative Myasthenia Gravis (QMG)
A physician assessment that quantifies disease severity. Measures are assessed on a scale of 0-3, with total scores ranging from 0 to 39.1
- Improvement of ≥3 points was established as clinically meaningful3
Measures include4:
- Ptosis
- Facial muscle weakness
- Dysarthria
- Grip strength
- Neck flexion endurance
- Diplopia
- Difficulty swallowing 4 oz of water
- Percentage predicted forced vital capacity
- Arm and leg endurance
Myasthenia Gravis Composite (MGC)
A 10-item patient and physician assessment of the signs and symptoms of MG, with total scores ranging from 0 to 50. Higher scores are interpreted as greater impairments.5
- Improvement of ≥3 points was established as clinically meaningful3
Measures include5:
- Ptosis
- Eye closure
- Chewing
- Breathing
- Shoulder abduction
- Diplopia
- Talking
- Swallowing
- Neck flexion/extension
- Hip flexion
Myasthenia Gravis Quality of Life 15-item revised (MG-QoL 15r) scale
A 15-item questionnaire that allows clinicians to estimate a patient’s quality of life relevant to MG. Items are scored from 0 (not at all) to 2 (very much). Cumulative scores range from 0 to 30, with higher scores representing worse quality of life.6
- Clinically meaningful was not defined for the MG-QoL 15r assessment3
Measures include6:
- Frustration
- Trouble eating
- Limitations at work
- Hobby and activity enjoyment
- Depression
- Mobility
- Personal grooming
- Loss of independence
RAISE-XT is an extension study of adult patients who opted to continue on ZILBRYSQ or switch to ZILBRYSQ from placebo. The primary objective was to evaluate the long-term safety and tolerability of ZILBRYSQ in study participants with generalized myasthenia gravis (gMG). Long-term efficacy was also studied through multiple measures as secondary endpoints.3
Additional RAISE-XT secondary endpoints
RAISE-XT: consistent improvements across key clinician- and patient-reported secondary and other outcome measures2‑5
ZILBRYSQ demonstrated sustained efficacy through Week E842,3
Secondary endpoint: CFB in the QMG total score at Week E12 (mITT population)3*
The long-term safety, tolerability, and efficacy were evaluated in an open-label extension study comprised of gMG patients who participated in either Phase 2 or Phase 3 (RAISE) of the clinical trials.3
RAISE-XT evaluated 200 patients from either Phase 2 or 3, including 17 patients from Phase 2 who started with or switched to 0.1 mg/kg. The graphic above shows only the 183 patients continuing or switching to ZILBRYSQ at the approved dosage (0.3 mg/kg) and are solely represented in the subsequent analyses on efficacy.2,3
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
*Includes pooled data of patients from Week E12-Week E84.2
CFB=change from baseline; CI=confidence interval; LS=least squares; mITT=modified intention-to-treat; SE=standard error.
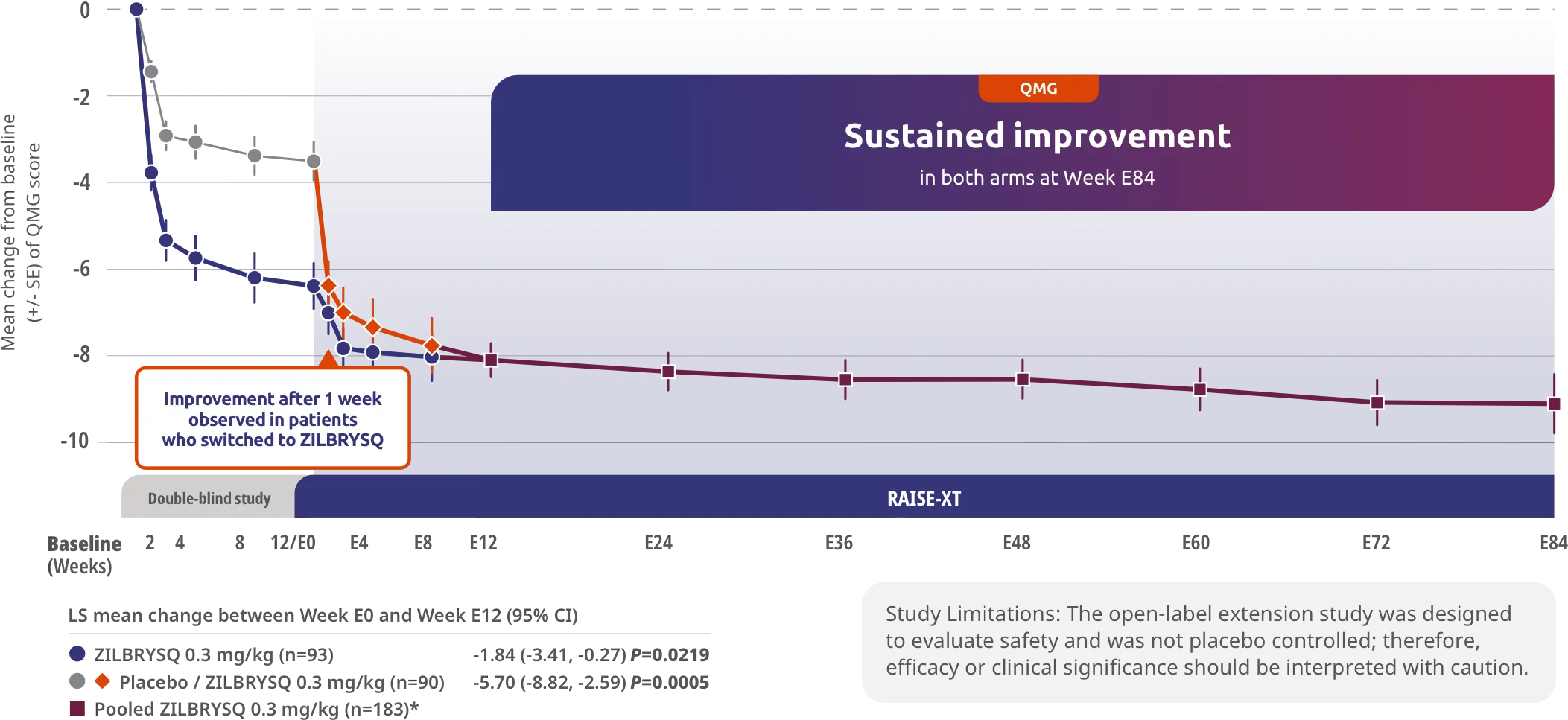
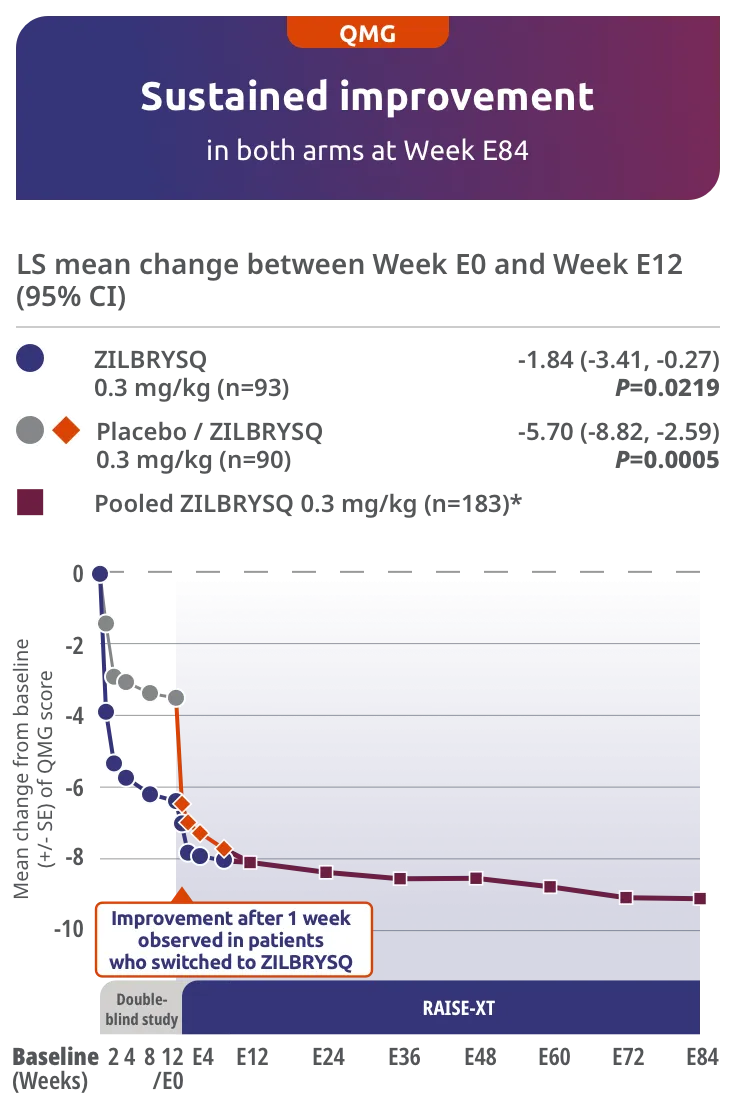
ZILBRYSQ demonstrated sustained efficacy through Week E842,3
Secondary endpoint: CFB in the MGC total score at Week E12 (mITT population)*
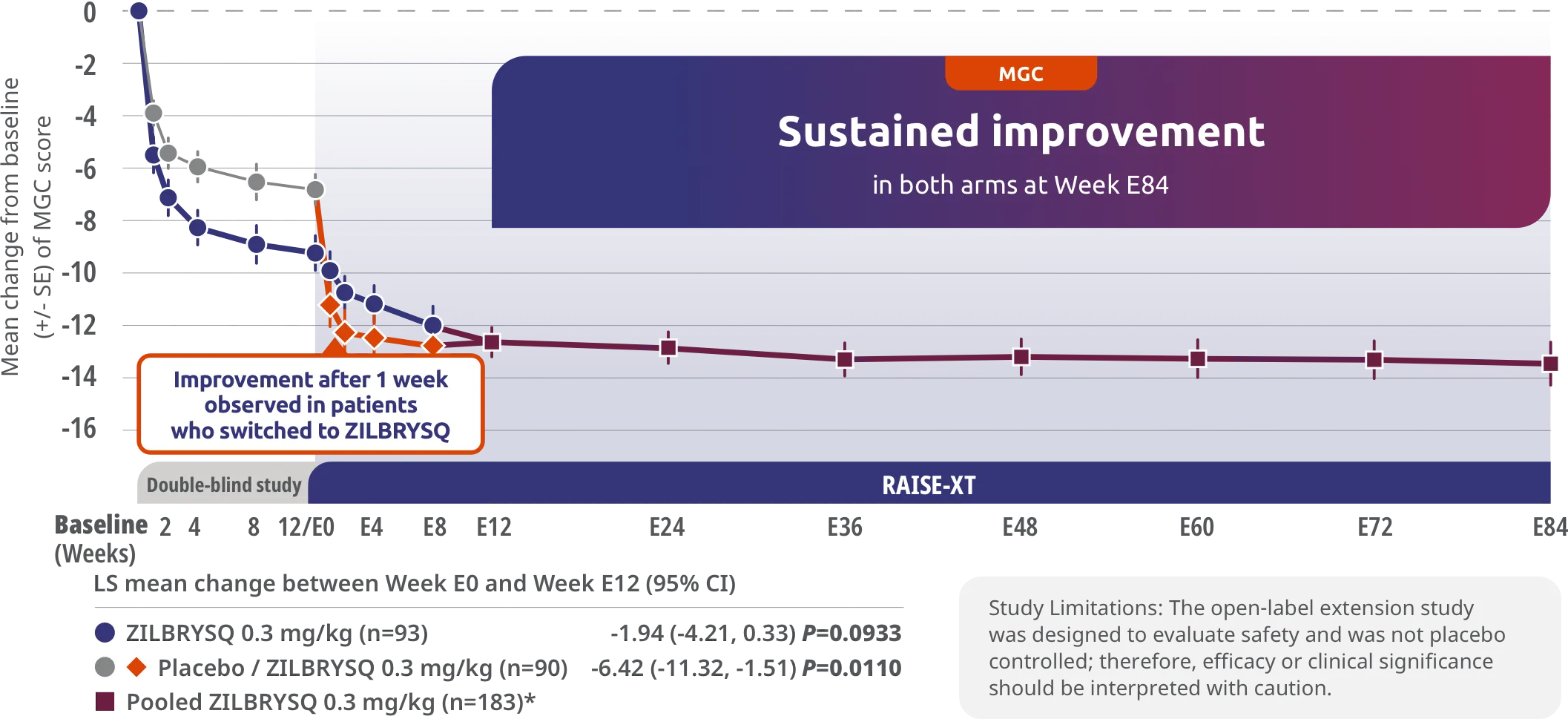
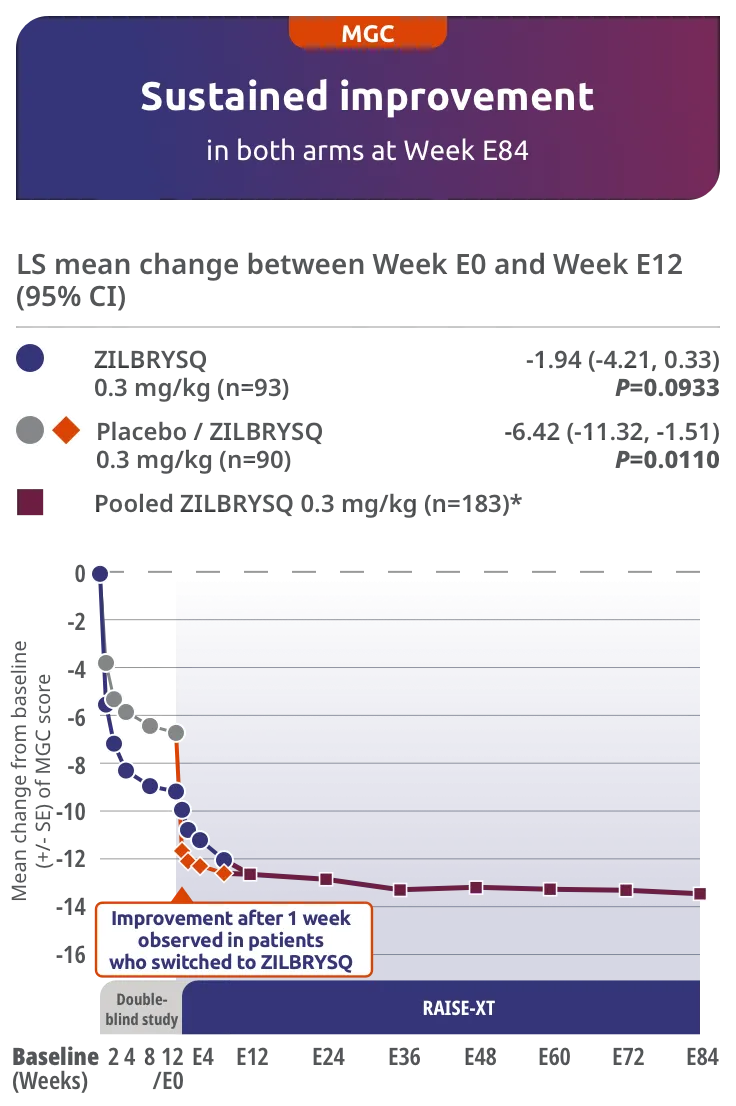
The long-term safety, tolerability, and efficacy were evaluated in an open-label extension study comprised of gMG patients who participated in either Phase 2 or Phase 3 (RAISE) of the clinical trials.3
RAISE-XT evaluated 200 adult patients from either Phase 2 or 3, including 17 patients from Phase 2 who started with or switched to 0.1 mg/kg. The graphic above shows only the 183 patients continuing or switching to ZILBRYSQ at the approved dosage (0.3 mg/kg) and are solely represented in the subsequent analyses on efficacy.2,3
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
*Includes pooled data of patients from Week E12-Week E84.2
CFB=change from baseline; CI=confidence interval; LS=least squares; mITT=modified intention-to-treat.
ZILBRYSQ demonstrated sustained efficacy through Week E842,3
Secondary endpoint: CFB in the MG-QoL 15r total score at Week E12 (mITT population)*
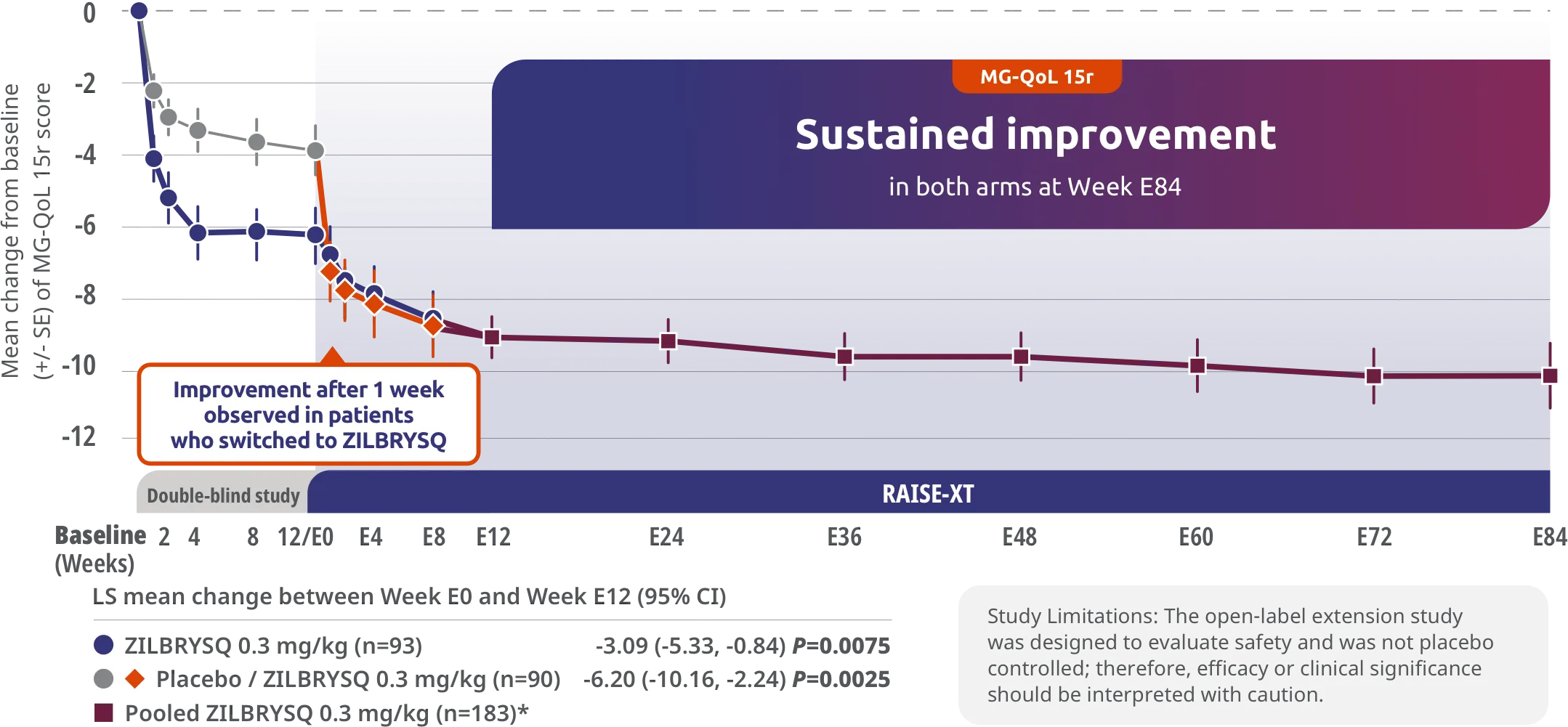
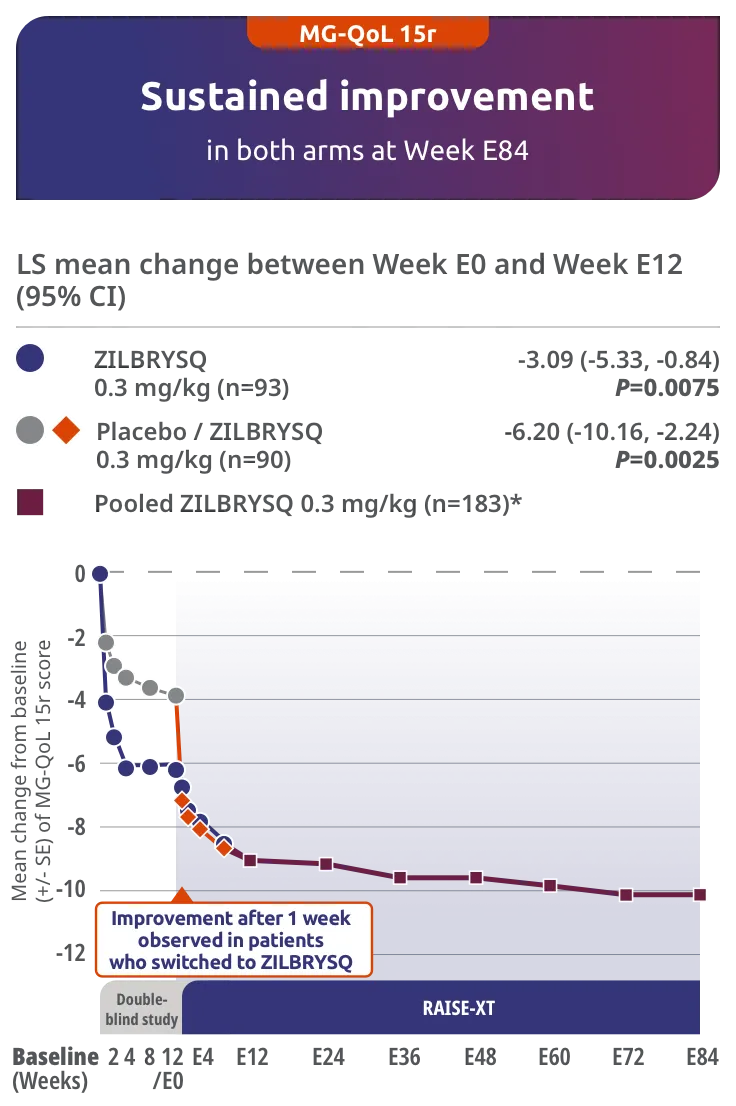
The long-term safety, tolerability, and efficacy were evaluated in an open-label extension study comprised of gMG patients who participated in either Phase 2 or Phase 3 (RAISE) of the clinical trials.3
RAISE-XT evaluated 200 adult patients from either Phase 2 or 3, including 17 patients from Phase 2 who started with or switched to 0.1 mg/kg. The graphic above shows only the 183 patients continuing or switching to ZILBRYSQ at the approved dosage (0.3 mg/kg) and are solely represented in the subsequent analyses on efficacy.2,3
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
*Includes pooled data of patients from Week E12-Week E84.2
CFB=change from baseline; CI=confidence interval; LS=least squares; mITT=modified intention-to-treat; SE=standard error.
RAISE-XT other endpoint:
Complement inhibition was achieved after Week 1 and sustained through Week E483*
Complement activity as shown by sRBC lysis assay
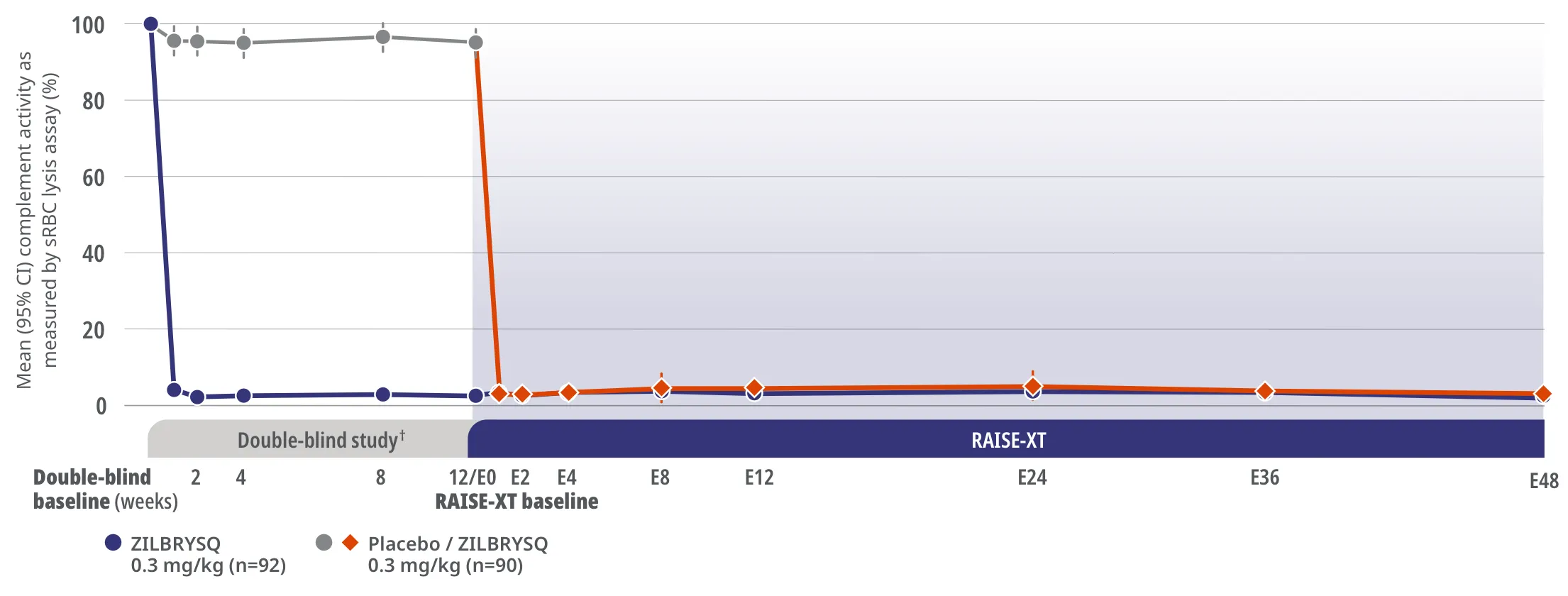
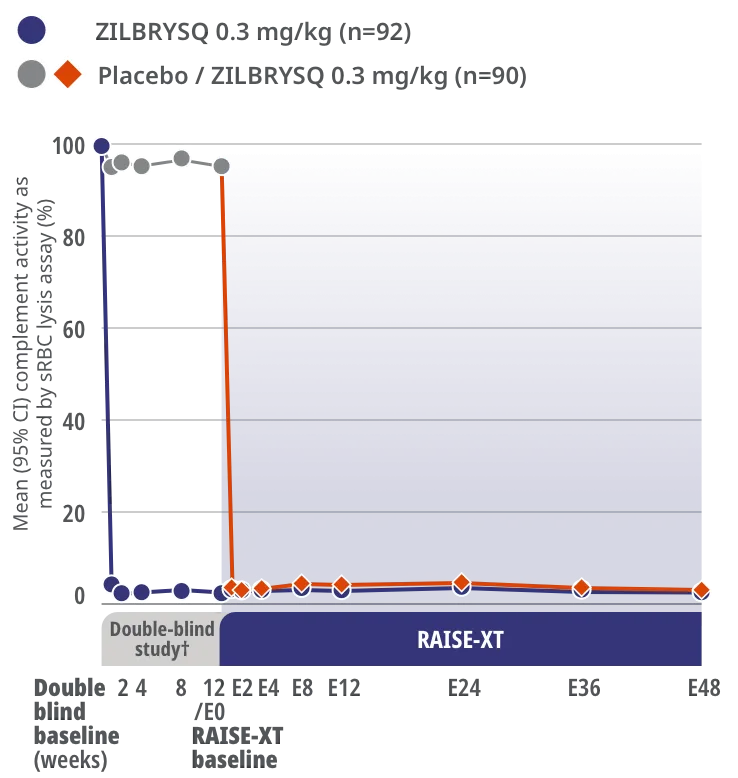
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
Assessment of complement activity using sRBC lysis assay was a pharmacodynamics outcome.3
In RAISE-XT, Week E48 was the data cut (September 8, 2022) for the assessment of complement activity.3
*RAISE-XT is an ongoing, multicenter, open-label extension study of ZILBRYSQ in study participants with anti-AChR Ab+ gMG who completed the Phase 2 or Phase 3 (RAISE) study (N=200). The analysis cutoff date was September 8, 2022.3
†The safety and efficacy of ZILBRYSQ were evaluated in RAISE, a 12-week, multicenter, randomized, double-blind, placebo-controlled study. The patient population included adult patients with a diagnosis of mild to severe gMG (MGFA class II-IV).1
Ab+=antibody positive; AChR=acetylcholine receptor; CI=confidence interval; MGFA=Myasthenia Gravis Foundation of America; sRBC=sheep red blood cell.
RAISE-XT post hoc data:
Post hoc analysis of secondary endpoint: use of rescue therapy in patients who switched from placebo to ZILBRYSQ in RAISE-XT5
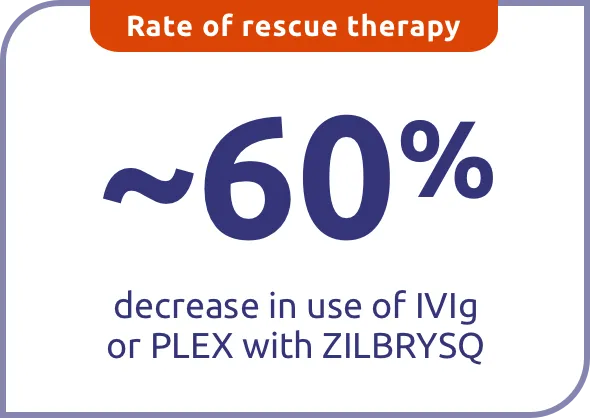
In RAISE, a 12-week, multicenter, randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of ZILBRYSQ, the cumulative proportion of patients receiving rescue therapy (other secondary efficacy outcome) by Week 12 was 12% for the placebo group (n=88) and 5% for the ZILBRYSQ group (n=86) (P=0.1003).3
Events per 100 patient-years*
Rate of rescue therapy use:
- 78.16 in patients taking placebo in double-blind period (n=103)
- 33.11 in patients switched from placebo to ZILBRYSQ in open-label extension (n=101)
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
The data was from a post hoc analysis based on a prespecified secondary endpoint not controlled for multiplicity and not powered; therefore, data should be interpreted with caution and conclusions cannot be drawn.
In RAISE, a 12-week, multicenter, randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of ZILBRYSQ, the cumulative proportion of patients receiving rescue therapy (other secondary efficacy outcome) by Week 12 was 12% for the placebo group (n=88) and 5% for the ZILBRYSQ group (n=86) (P=0.1003).3
Patients who switched from placebo to ZILBRYSQ in RAISE-XT had ~60% decrease in utilization of IVIg and PLEX compared to the double-blind period at the time of data cutoff (September 8, 2022) (median exposure 1.2 [range: 0.11-4.45] years).*
*Double-blind period: Weeks 0-12. The rate of rescue therapy use during the double-blind period was adjusted for exposure.
IVIg=intravenous immunoglobulin; PLEX=plasma exchange.
RAISE-XT post hoc analysis: change in corticosteroid dose at Week E485*
Exploratory endpoint: proportion of patients with reduced or discontinued corticosteroid dose compared to double-blind baseline†
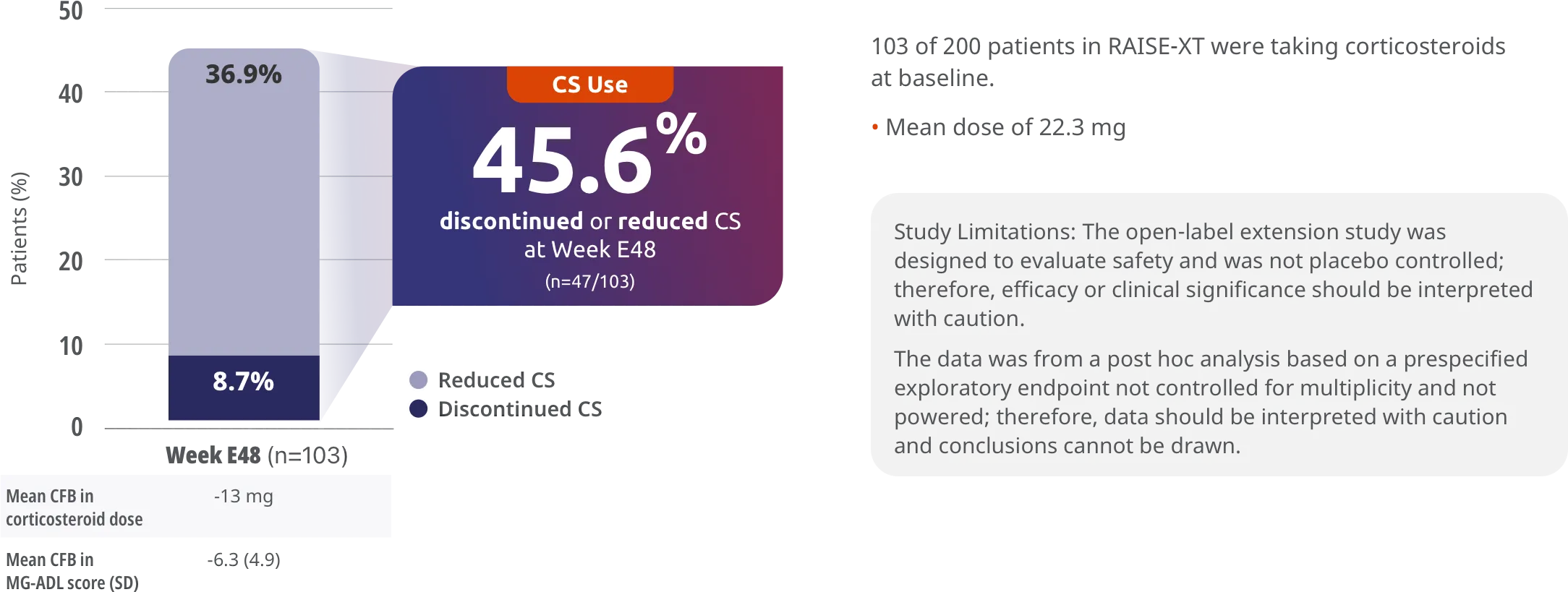
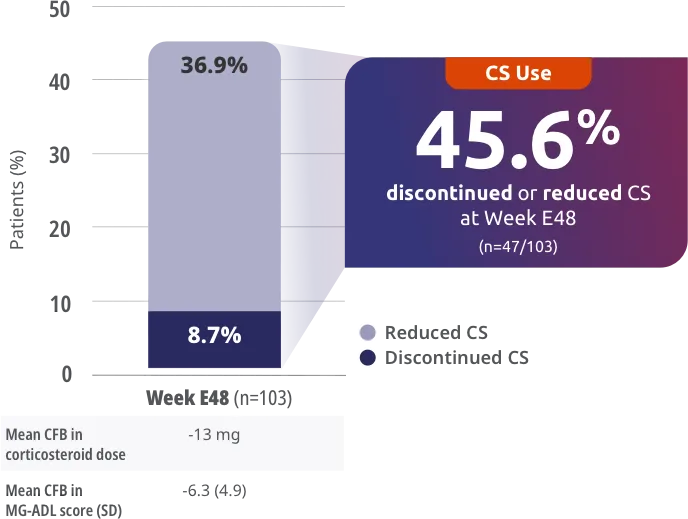
103 of 200 patients in RAISE-XT were taking corticosteroids at baseline.
- Mean dose of 22.3 mg
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
The data was from a post hoc analysis based on a prespecified exploratory endpoint not controlled for multiplicity and not powered; therefore, data should be interpreted with caution and conclusions cannot be drawn.
Week E48 was the first data cut (September 8, 2022) for the post hoc interim analysis of change in corticosteroid dose.3,5 Additional data cuts will be presented in the future from the open-label extension study.
*RAISE-XT is an ongoing, multicenter, open-label extension study of ZILBRYSQ in study participants with anti-AChR Ab+ gMG who completed the Phase 2 or Phase 3 (RAISE) study (N=200). The analysis cutoff date was September 8, 2022.3
†Analysis included patients with >0 mg corticosteroid dose at baseline in the double-blind study. Corticosteroid dose remained stable prior to Week E12. The analysis cutoff date was November 11, 2023.5
Ab+=antibody positive; AChR=acetylcholine receptor; CFB=change from baseline; CS=corticosteroid; MG-ADL=Myasthenia Gravis Activities of Daily Living; SD=standard deviation.
Post hoc analysis of exploratory endpoint: complement inhibition was maintained during concomitant use of ZILBRYSQ with IVIg and PLEX3,6
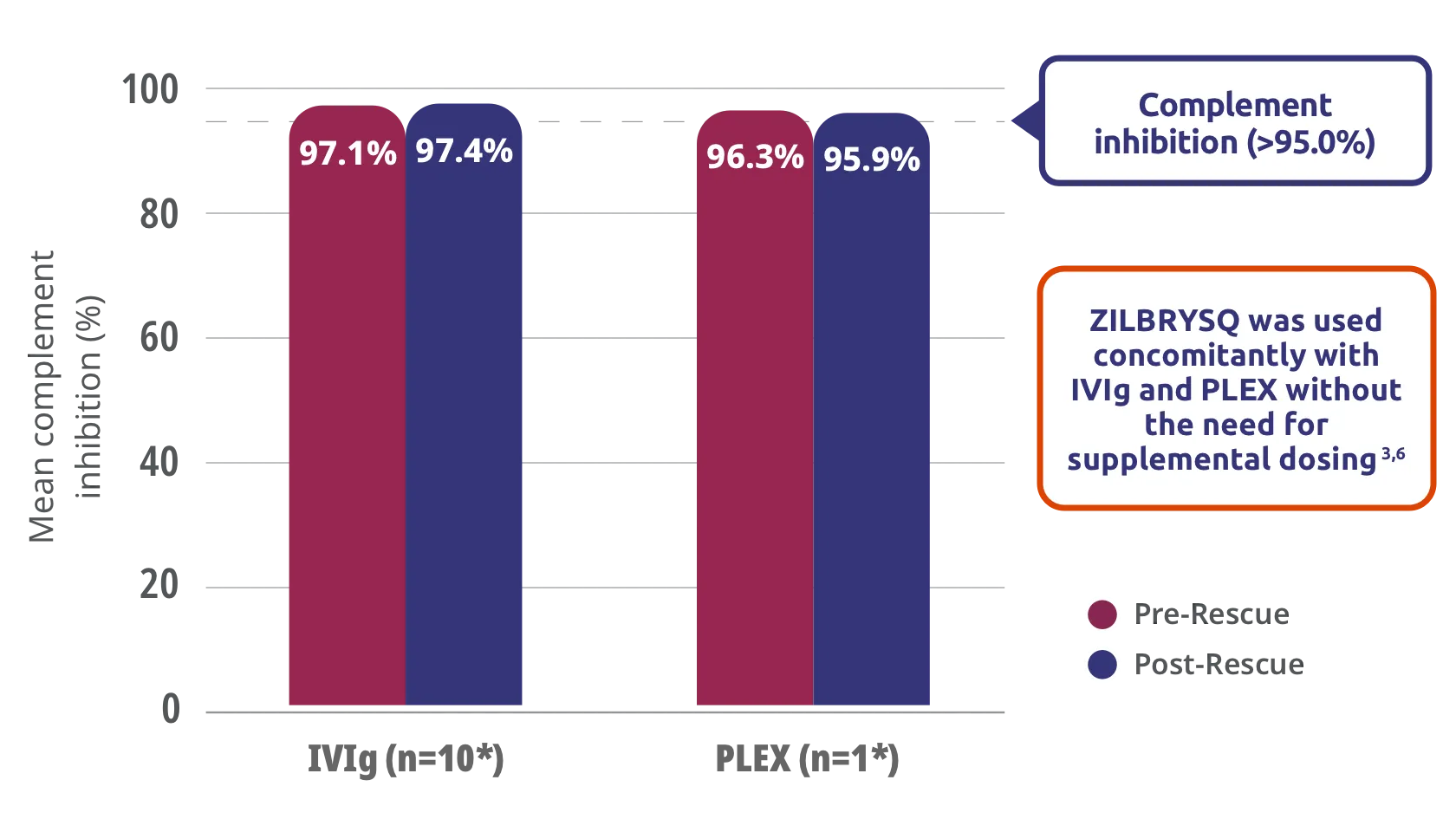
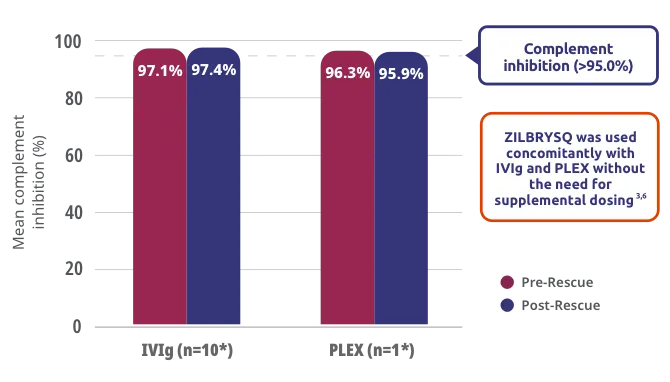
Study Limitations: The open-label extension study was designed to evaluate safety and was not placebo controlled; therefore, efficacy or clinical significance should be interpreted with caution.
The data was from a post hoc analysis based on a prespecified exploratory endpoint not controlled for multiplicity and not powered; therefore, data should be interpreted with caution and conclusions cannot be drawn.
Assessment of complement activity using sRBC lysis assay was a pharmacodynamics outcome.3,6
*Events with available data. Complement activity was measured by sRBC lysis assay, with post-administration measurement taken up to 1 day after rescue treatment. Data cutoff: September 8, 2022.6
RAISE-XT is an ongoing, multicenter, open-label extension study of ZILBRYSQ in adult study participants with anti-AChR Ab+ gMG who completed the Phase 2 or Phase 3 (RAISE) study (N=200). The analysis cutoff date was September 8, 2022.6
IVIg=intravenous immunoglobulin; PLEX=plasma exchange; sRBC=sheep red blood cell.
RAISE-XT secondary endpoint measures overview
Quantitative Myasthenia Gravis (QMG)
A physician assessment that quantifies disease severity. Measures are assessed on a scale of 0-3, with total scores ranging from 0 to 39.1
- Improvement of ≥3 points was established as clinically meaningful4
Measures include8:
- Ptosis
- Facial muscle weakness
- Dysarthria
- Grip strength
- Neck flexion endurance
- Diplopia
- Difficulty swallowing 4 oz of water
- Percentage predicted forced vital capacity
- Arm and leg endurance
Myasthenia Gravis Composite (MGC)
A 10-item patient and physician assessment of the signs and symptoms of MG, with total scores ranging from 0 to 50. Higher scores are interpreted as greater impairments.8
- Improvement of ≥3 points was established as clinically meaningful4
Measures include8:
- Ptosis
- Eye closure
- Chewing
- Breathing
- Shoulder abduction
- Diplopia
- Talking
- Swallowing
- Neck flexion/extension
- Hip flexion
Myasthenia Gravis Quality of Life 15-item revised (MG-QoL 15r) scale
A 15-item questionnaire that allows clinicians to estimate a patient’s quality of life relevant to MG. Items are scored from 0 (not at all) to 2 (very much). Cumulative scores range from 0 to 30, with higher scores representing worse quality of life.9
- Clinically meaningful was not defined for the MG-QoL 15r assessment4
Measures include9:
- Frustration
- Trouble eating
- Limitations at work
- Hobby and activity enjoyment
- Depression
- Mobility
- Personal grooming
- Loss of independence


References:
- ZILBRYSQ [Prescribing Information]. Smyrna, GA: UCB, Inc.
- Leite MI, Bresch S, Hewamadduma C, et al; on behalf of the RAISE-XT Study Team. Long-term zilucoplan in generalised myasthenia gravis: 96-week follow-up interim analysis of RAISE-XT. Presented at: European Academy of Neurology 2024 Annual Meeting; April 13-18, 2024; Helsinki, Finland. Presentation EPR-254.
- Howard JF Jr, Bresch S, Genge A, et al; RAISE Study Team. Safety and efficacy of zilucoplan in patients with generalised myasthenia gravis (RAISE): a randomised, double-blind, placebo-controlled, Phase 3 study. Lancet Neurol. 2023;22(5):395-406. doi:10.1016/S1474-4422(23)00080-7
- Conquer MG. MG Activities of Daily Living (MG-ADL) scale. September 29, 2022. Accessed November 9, 2023. https://www.myastheniagravis.org/mg-activities-of-daily-living-mg-adl-scale/
- Howard JF Jr, Bresch S, Farmakidis C, et al. Long-term safety and efficacy of zilucoplan in patients with generalized myasthenia gravis: interim analysis of the RAISE-XT open-label extension study. Ther Adv Neurol Disord. 2024;17(3):1-16. doi:10.1177/17562864241243186
- Vu T, Genge A, Hussain Y, et al; on behalf of the RAISE Investigators. Efficacy and safety of zilucoplan in myasthenia gravis: responder analysis from the randomized Phase 3 RAISE trial. Poster presented at: American Association of Neuromuscular & Electrodiagnostic Medicine Annual Meeting; September 21-24, 2022; Nashville, TN. Poster 200.
- ZILBRYSQ [Prescribing Information]. Smyrna, GA: UCB, Inc.
- Leite MI, Bresch S, Hewamadduma C, et al; on behalf of the RAISE-XT Study Team. Long-term zilucoplan in generalised myasthenia gravis: 96-week follow-up interim analysis of RAISE-XT. Presented at: European Academy of Neurology 2024 Annual Meeting; April 13-18, 2024; Helsinki, Finland. Presentation EPR-254.
- Howard JF Jr, Bresch S, Genge A, et al; RAISE Study Team. Safety and efficacy of zilucoplan in patients with generalised myasthenia gravis (RAISE): a randomised, double-blind, placebo-controlled, Phase 3 study. Lancet Neurol. 2023;22(5):395-406. doi:10.1016/S1474-4422(23)00080-7
- QMG form. Myasthenia Gravis Foundation of America. 1997. Accessed November 9, 2023. https://myasthenia.org/Portals/0/QMG.pdf
- Sadjadi R, Conaway M, Cutter G, et al; MG Composite MG-QOL15 Study Group. Psychometric evaluation of the myasthenia gravis composite using Rasch analysis. Muscle Nerve. 2012;45(6):820-825. doi:10.1002/mus.23260
- Myasthenia Gravis Quality of Life-15 Revised (MG-QOL15R). Myasthenia Gravis Rare Disease Network. September 7, 2022. Accessed October 25, 2023. https://mgnet.rarediseasesnetwork.org/sites/default/files/2023-04/mg-quality-life-15-revised.pdf
- ZILBRYSQ [Prescribing Information]. Smyrna, GA: UCB, Inc.
- Leite MI, Bresch S, Hewamadduma C, et al; on behalf of the RAISE-XT Study Team. Long-term zilucoplan in generalised myasthenia gravis: 96-week follow-up interim analysis of RAISE-XT. Presented at: European Academy of Neurology 2024 Annual Meeting; April 13-18, 2024; Helsinki, Finland. Presentation EPR-254.
- Howard JF Jr, Bresch S, Farmakidis C, et al. Long-term safety and efficacy of zilucoplan in patients with generalized myasthenia gravis: interim analysis of the RAISE-XT open-label extension study. Ther Adv Neurol Disord. 2024;17(3):1-16. doi:10.1177/17562864241243186
- Howard JF Jr, Bresch S, Genge A, et al; RAISE Study Team. Safety and efficacy of zilucoplan in patients with generalised myasthenia gravis (RAISE): a randomised, double-blind, placebo-controlled, Phase 3 study. Lancet Neurol. 2023;22(5):395-406. doi:10.1016/S1474-4422(23)00080-7
- Hewamadduma C, Genge A, Freimer M, et al; on behalf of the RAISE-XT Study Team. Corticosteroid dose tapering in patients with generalised myasthenia gravis on zilucoplan: interim analysis of RAISE-XT. Poster presented at: Academy of British Neurologists Annual Meeting; May 21-23, 2024; Edinburgh, UK. Poster 183.
- Howard JF Jr, Freimer M, Genge A, et al; on behalf of the RAISE-XT Study Team. Long-term safety and efficacy of zilucoplan in myasthenia gravis: additional interim analyses of RAISE-XT. Presented at: American Academy of Neurology 2024 Annual Meeting; April 13-18, 2024; Denver, CO. Presentation S15.002.
- QMG form. Myasthenia Gravis Foundation of America. 1997. Accessed November 9, 2023. https://myasthenia.org/Portals/0/QMG.pdf
- Sadjadi R, Conaway M, Cutter G, et al; MG Composite MG-QOL15 Study Group. Psychometric evaluation of the myasthenia gravis composite using Rasch analysis. Muscle Nerve. 2012;45(6):820-825. doi:10.1002/mus.23260
- Myasthenia Gravis Quality of Life-15 Revised (MG-QOL15R). Myasthenia Gravis Rare Disease Network. September 7, 2022. Accessed October 25, 2023. https://mgnet.rarediseasesnetwork.org/sites/default/files/2023-04/mg-quality-life-15-revised.pdf


